We investigate relationship between autophagy and DNA damage repair, to optimize curative therapeutic strategies
Genomic instability is the main etiological factor for carcinogenesis and plays a critical role in neurodegenerative diseases and aging. Cells have evolved complex mechanisms to preserve genome and, when environmental and endogenous DNA damage-inducing agents threaten genomic stability, DNA damage response (DDR) is activated, leading to the detection, signaling and repair of lesions or, if damage is irreparable, to senescence or cell death.
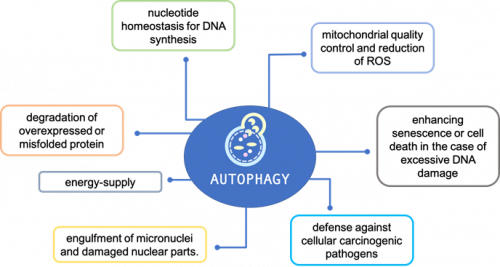
Autophagy and genome maintenance
A range of mechanisms appear to be involved where autophagy participates in preventing genomic instability, as well as in DDR and cell fate decision (Figure 1).
However, the crosstalk between autophagy and DDR is just beginning to be explored and key questions remain that need to be addressed. First, precisely, how is double strand break (DSB) repair regulated by autophagy, and are these mechanisms of regulation altered in the context of cancer? Second, what dictates the impact of autophagy modulation on cancer cell fate?
The aim of our group is to understand how autophagy influences DSB repair pathways
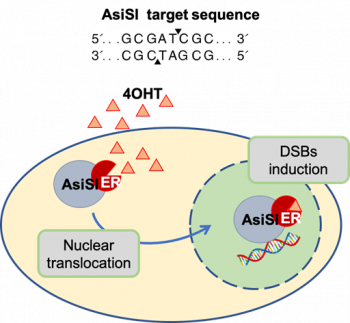
We transduced the MCF10A cells with a retroviral vector expressing the fusion protein between the HA-tagged AsiSI restriction enzyme and a modified hormone-binding domain from the estrogen receptor. Cell exposure to 4-hydroxytamoxifen (4OHT) results in nuclear accumulation of the AsiSI fusion protein and in the rapid induction of approximately 150 sequence–specific DSBs across the genome (Figure 2). Based on our previous studies in U2OS and MCF10A, AsiSI dependent DSBs induce canonical DDR activation, which is followed by progressive resolution of DSBs.
Our cellular system provides a unique opportunity to simultaneously study, using ChIP-seq and PCR based approaches, the repair events that transpire at many different lesions located within various chromatin states.
These studies will provide important information about autophagy targeting in the context of cancer, age-related pathologies and neurodegeneration.
Autophagy and resistance to therapy in cancer
Resistance to chemotherapy represents a major clinical problem for the management of cancer patients. Since it is known that autophagy is stimulated upon exposure to diverse DNA damaging factors, this poses problems for many forms of chemotherapy, leading to drug resistance. Although many lines of evidence suggest the feasibility of autophagy as a target in anticancer therapy, results may be viewed as controversial so far. Although one possible explanation is that the functional consequences of autophagy may be different according to tissue of origin and stage of progression, there are likely to be as yet undefined bio-chemical and/or molecular factors that dictate the impact of autophagy on cell fate.
The use of integrative omics and in vivo studies provides a great opportunity to reach a full understanding of the complex and intricate relationship between autophagy and chemo-resistance and could have profound impacts on our ability to treat cancer.
The aim of our group is to assess the clinical elevance of autophagy in resistance to therapy, by in vivo umor phenotyping experiments, along with bioinformatics approaches for the identification of effectors candidates and biomarkers.
We expect to generate instrumental information on how autophagy participates in development of resistance to therapy in cancer.